Pronunciation: EN-still-ar
Generic name: calcipotriene and betamethasone dipropionate
Dosage form: foam
Drug class: Topical antipsoriatics
What is Enstilar?
Enstilar foam is a once-daily, topical, fixed-dose combination foam containing calcipotriene 0.005% (a vitamin D analog) and betamethasone dipropionate 0.064% (a corticosteroid) that may be used to treat plaque psoriasis in adults and children 12 years of age or older.
Enstilar foam is a stable supersaturated solution of calcipotriol/betamethasone dipropionate that has improved clinical efficacy and increased skin penetration compared with standard calcipotriol/betamethasone dipropionate ointments.
Enstilar was approved on October 16, 2015.
Warnings
Use only as directed. Tell your doctor if you use other medicines or have other medical conditions or allergies.
Hypercalcemia and hypercalciuria have been observed with the use of topical calcipotriene. If either occurs, discontinue Enstilar foam until laboratory values of calcium normalize.
Endocrine effects
The use of high-potency steroid products such as betamethasone dipropionate can cause reversible hypothalamic-pituitary-adrenal (HPA) axis suppression with the potential for glucocorticosteroid insufficiency during and after withdrawal of treatment. Risk factors include the use of high-potency topical corticosteroids, use over a large surface area, or to areas under occlusion, prolonged use, altered skin barrier, liver failure, and young age. Modify use of Enstilar foam should HPA axis suppression develop.
Ophthalmic Adverse Reactions
May increase the risk of cataracts and glaucoma. If visual symptoms occur, consider referral to an ophthalmologist.
Allergic contact dermatitis
May occur with either calcipotriene or betamethasone dipropionate topical use. May be observed as a failure to heal. Confirm with diagnostic patch testing.
Children
Children can absorb larger amounts of this medicine through the skin and may be more likely to have side effects. Enstilar foam is not approved for use by anyone younger than 12 years old.
Before taking this medicine
You should not use Enstilar foam if you are allergic to calcipotriene, betamethasone dipropionate, or any other components of the foam.
Tell your doctor if you have ever had:
- too much calcium in your blood or urine
- an adrenal gland disorder
- cataracts or glaucoma
- liver disease
- severe forms of psoriasis (with pus, skin peeling, severe redness)
- have thinning skin at the treatment site
- a skin infection or
- if you are receiving UV light treatments (phototherapy).
Pregnancy
The use of high-potency topical steroid medicine during pregnancy may increase the risk of a low birth weight in the newborn baby. Tell your doctor if you are pregnant or plan to become pregnant.
Lactation
It may not be safe to breastfeed while using Enstilar. Ask your doctor about any risks. If you apply betamethasone and calcipotriene to your chest, avoid areas that may come into contact with the baby’s mouth.
How is Enstilar foam used?
Enstilar should be applied once daily to the plaques for up to 4 weeks.
- Shake the can before applying the foam.
- Hold the can 1.5 inches (one and a half inches) away from the affected areas and spray the foam onto the plaques by pushing down on the nozzle, in any position except sideways.
- Gently rub in the foam until it is absorbed. After applying, put the cap back on the can and you’re your hands.
- Discontinue treatment once control has been achieved.
- Do not use more than 60 grams every 4 days.
- Do not apply an occlusive dressing over the top of Enstilar foam unless directed by a physician.
- Do not use it on your face, groin, or armpit, or if skin thinning is present at the treatment site.
- Not for oral, ophthalmic, or intravaginal use.
The contents of the Enstilar can are flammable and you should avoid being near sources of fire or flames, or smoking, immediately following application.
What are the side effects of Enstilar?
The most common side effects reported with Enstilar include irritation or itch at the application site, folliculitis, skin hypopigmentation, high calcium levels, urticaria, and exacerbation of psoriasis.
Postmarketing studies with topical corticosteroids have reported atrophy, striae, telangiectasias, itching, dryness, hypopigmentation, perioral dermatitis, secondary infection, and miliaria.
Ophthalmic adverse reactions of cataracts, glaucoma, and increased intraocular pressure, have been reported during the use of topical corticosteroids, including topical betamethasone products.
What happens if I miss a dose?
Apply Enstilar foam as soon as you can, but skip the missed dose if it is almost time for your next dose. Do not apply two doses at one time.
What happens if I overdose?
Seek emergency medical attention or call the Poison Help line at 1-800-222-1222 if anyone has accidentally swallowed the medicine.
High doses or long-term use of Enstilar can lead to thinning skin, easy bruising, changes in body fat (especially in your face, neck, back, and waist), increased acne or facial hair, menstrual problems, impotence, or loss of interest in sex.
What should I avoid while using Enstilar?
Avoid using other skin medications that contain betamethasone or calcipotriene. Do not use Enstilar to treat any skin condition that has not been checked by your doctor.
Avoid getting the foam in your eyes or on your face, underarms, groin area, or areas of thin skin.
Enstilar foam could make you sunburn more easily. Avoid sunlight or tanning beds. Wear protective clothing and use sunscreen (SPF 30 or higher) when you are outdoors.
Get emergency medical help if you have signs of an allergic reaction: hives; difficulty breathing; swelling of your face, lips, tongue, or throat.
Side effects can occur if your skin absorbs topical steroid medicine. Tell your doctor if you have:
- swelling, weight gain (especially in your face or your upper back and torso);
- thinning skin, stretch marks, acne, increased facial hair;
- irregular menstrual periods, changes in sexual function; or
- memory problems, and feeling irritable.
Also, tell your doctor if you have:
- worsening of your skin condition;
- redness, warmth, swelling, oozing, or severe irritation of any treated skin;
- blurred vision, tunnel vision, eye pain, or seeing halos around lights;
- high calcium levels—confusion, tiredness, nausea, vomiting, loss of appetite, constipation, increased thirst or urination, weight loss; or
- high blood sugar—increased thirst, increased urination, dry mouth, fruity breath odor.
What other drugs will affect Enstilar?
Tell your doctor about all your other medicines, especially:
- any other steroid medicine (such as dexamethasone, fludrocortisone, hydrocortisone, prednisone, triamcinolone, and others); or
- any other medicine to treat psoriasis.
This list is not complete. Other drugs may affect Enstilar foam, including prescription and over-the-counter medicines, vitamins, and herbal products. Not all possible drug interactions are listed.
Ingredients
Active ingredients: Each gram of Enstilar foam contains 52.2 mcg of calcipotriene hydrate (equivalent to 50 mcg of calcipotriene) and 0.643 mg of betamethasone dipropionate (equivalent to 0.5mg of betamethasone). It is available in 60-gram aluminum cans.
Inactive ingredients: white petrolatum PPG-11 stearyl ether, mineral oil, all-rac-alpha-tocopherol, and butylhydroxytoluene.
Propellants: dimethyl ether and butane.
Storage
Store between 20°C-25°C (68°F-77°F) and keep away from heat. Do not expose the can to a heat source or store it at temperatures above 49°C (120°F)
Do not puncture or burn the can. Do not freeze.
Do not use past the expiry date printed on the side of the can.
Discard any unused product 6 months after the can has been opened.
Keep out of reach of children.
Manufacturer
Leo Pharma.
Popular FAQ
Enstilar is a topical spray foam applied to the skin to treat plaque psoriasis. It contains calcipotriene, a vitamin D analog, and betamethasone dipropionate, a potent corticosteroid. Enstilar is used on the skin for up to 4 weeks in people 12 years and older. Continue reading
In studies, Enstilar Foam (calcipotriene / betamethasone dipropionate) has been used safely and effectively to treat plaque psoriasis on the body and scalp in adults and adolescents ages 12 to 17 years of age. Foam preparations are often preferred by patients with scalp psoriasis (over ointments or thick creams) and the ingredients in Enstilar Foam are considered standard first-line treatments. Continue reading
Further information
Always consult your healthcare provider to ensure the information displayed on this page applies to your personal circumstances.
Medical Disclaimer
Country: United Kingdom
Language: English
Source: MHRA (Medicines & Healthcare Products Regulatory Agency)
Buy It Now
Active ingredient:
Betamethasone dipropionate; Calcipotriol monohydrate
Available from:
LEO Pharma
ATC code:
D05AX52
INN (International Name):
Betamethasone dipropionate; Calcipotriol monohydrate
Dosage:
500microgram/1gram ; 50microgram/1gram
Pharmaceutical form:
Foam
Administration route:
Cutaneous
Class:
No Controlled Drug Status
Prescription type:
Valid as a prescribable product
Product summary:
BNF: 13050200; GTIN: 5702191012125 5702191015799
Patient Information leaflet
BACK
FRONT
000000-00
420 mm
287 mm
NO VISION CONTROL UNTIL FURTHER NOTICE!
JUG 27.06.2014
WHAT IS IN THIS LEAFLET
1. What Enstilar
®
is and what it is used for
2. What you need to know before you use Enstilar
®
3. How to use Enstilar
®
4. Possible side effects
5. How to store Enstilar
®
6. Contents of the pack and other information
1. WHAT ENSTILAR
®
IS AND WHAT IT IS USED FOR
Enstilar is used on the skin to treat psoriasis vulgaris in
adults. Psoriasis is caused by your skin cells being produced
too quickly. This causes redness, scaling and thickness of
your skin.
Enstilar contains calcipotriol and betamethasone.
Calcipotriol helps to bring the rate of skin cell growth back to
normal and betamethasone helps to reduce the inflammation.
2. WHAT YOU NEED TO KNOW BEFORE YOU USE ENSTILAR
®
DO NOT USE ENSTILAR:
• if you are allergic to calcipotriol, betamethasone or any of
the other ingredients of this medicine (listed in section 6)
• if you have problems with calcium levels in your blood
(ask your doctor)
• if you have certain types of psoriasis called: erythrodermic
psoriasis or pustular psoriasis (ask your doctor if you are
unsure).
AS ENSTILAR CONTAINS A STRONG STEROID, DO NOT USE ENSTILAR
ON SKIN AREAS AFFECTED BY:
• skin infections caused by viruses (e.g. cold sores or
chicken pox)
• skin infections caused by a fungus (e.g. athlete’s foot and
ringworm)
• skin infections caused by bacteria
• skin infections caused by parasites (e.g. scabies)
• tuberculosis (TB)
• perioral dermatitis (red rash around the mouth)
• thin skin, easily damaged veins, stretch marks
• ichthyosis (dry skin with fish-like scales)
• acne (pimples)
• rosacea (severe flushing or redness of the skin on the face)
• ulcers and wounds.
WARNINGS AND PRECAUTIONS
TALK TO YOUR DOCTOR, PHARMACIST OR NURSE BEFORE USING
ENSTILAR IF:
• you have diabetes mellitus (diabetes), as your blood sugar
level may be affected by the steroid
• you are using other medicines that contain steroids, as
you may get side effects
•
Read the complete document
Summary of Product characteristics
OBJECT 1
ENSTILAR CUTANEOUS FOAM
Summary of Product Characteristics Updated 10-May-2016 | Leo
Laboratories Limited
1. Name of the medicinal product
Enstilar 50 micrograms/g + 0.5 mg/g cutaneous foam
2. Qualitative and quantitative composition
One gram of cutaneous foam contains 50 micrograms of calcipotriol (as
monohydrate) and 0.5 mg of
betamethasone (as dipropionate).
Excipient with known effect:
Butylhydroxytoluene (E321) 50 micrograms/g cutaneous foam.
For the full list of excipients, see section 6.1.
3. Pharmaceutical form
Cutaneous foam.
After spraying, a white to off-white foam is formed.
4. Clinical particulars
4.1 Therapeutic indications
Topical treatment of psoriasis vulgaris in adults.
4.2 Posology and method of administration
Posology
Enstilar foam should be applied to the affected area once daily. The
recommended treatment period is 4
weeks. The daily maximum dose of Enstilar should not exceed 15 g, i.e.
one 60 g can should last for at
least 4 days. 15 g corresponds to the amount administered from the can
if the actuator is fully depressed
for approximately one minute. A two-second application delivers
approximately 0.5 g. As a guide, 0.5 g
of foam should cover an area of skin roughly corresponding to the
surface area of an adult hand.
If using other topical products containing calcipotriol in addition to
Enstilar, the total dose of all
calcipotriol containing products should not exceed 15 g per day.
The total body surface area treated should not exceed 30%.
Special populations
_Renal and hepatic impairment_
The safety and efficacy of Enstilar foam in patients with severe renal
insufficiency or severe hepatic
disorders have not been evaluated.
_Paediatric population_
The safety and efficacy of Enstilar foam in children below 18 years
have not been established. No data
are available.
Method of administration
For cutaneous use.
The can should be shaken for a few seconds before use. Enstilar should
be applied by spraying holding
the can at least 3 cm from the skin. The foam can be sprayed holding
the ca
Read the complete document
Similar products
Search alerts related to this product
View documents history
Package leaflet: Information for the patient
Enstilar® 50 micrograms/g + 0.5 mg/g cutaneous foam
calcipotriol/betamethasone
Read all of this leaflet carefully before you start using this medicine because it contains important information for you.
• Keep this leaflet. You may need to read it again.
• If you have any further questions, ask your doctor, pharmacist or nurse.
• This medicine has been prescribed for you only. Do not pass it on to others. It may harm them, even if their signs of illness are the same as yours.
• If you get any side effects, talk to your doctor, pharmacist or nurse. This includes any possible side effects not listed in this leaflet. See section 4.
What is in this leaflet
1. What Enstilar® is and what it is used for
2. What you need to know before you use Enstilar®
3. How to use Enstilar®
4. Possible side effects
5. How to store Enstilar®
6. Contents of the pack and other information
1. What Enstilar® is and what it is used for
Enstilar is used on the skin to treat psoriasis vulgaris in adults. Psoriasis is caused by your skin cells being produced too quickly. This causes redness, scaling and thickness of your skin.
Enstilar contains calcipotriol and betamethasone.
Calcipotriol helps to bring the rate of skin cell growth back to normal and betamethasone helps to reduce the inflammation.
2. What you need to know before you use Enstilar®
Do not use Enstilar:
• if you are allergic to calcipotriol, betamethasone or any of the other ingredients of this medicine (listed in section 6)
• if you have problems with calcium levels in your blood (ask your doctor)
• if you have certain types of psoriasis called: erythrodermic psoriasis or pustular psoriasis (ask your doctor if you are unsure).
As Enstilar contains a strong steroid, do NOT use Enstilar on skin areas affected by:
• skin infections caused by viruses (e.g. cold sores or chicken pox)
• skin infections caused by a fungus (e.g. athlete’s foot and ringworm)
• skin infections caused by bacteria
• skin infections caused by parasites (e.g. scabies)
• tuberculosis (TB)
• perioral dermatitis (red rash around the mouth)
• thin skin, easily damaged veins, stretch marks
• ichthyosis (dry skin with fish-like scales)
• acne (pimples)
• rosacea (severe flushing or redness of the skin on the face)
• ulcers and wounds.
Warnings and precautions
Talk to your doctor, pharmacist or nurse before using Enstilar if:
• you have diabetes mellitus (diabetes), as your blood sugar level may be affected by the steroid
• you are using other medicines that contain steroids, as you may get side effects
• you have a certain type of psoriasis called guttate psoriasis.
Talk to your doctor, pharmacist or nurse during treatment if:
• you have used this medicine for a long period of time and plan to stop (as there is a risk your psoriasis will get worse or ‘flare up’ when steroids are stopped suddenly)
• your skin becomes infected, as you may need to stop your treatment
• the calcium level in your blood changes (see section 4 for further information).
Special precautions:
• avoid use under bandages or dressings as this increases the absorption of the steroid
• avoid use of more than 15 grams per day. This means that one 60 g can of Enstilar should last for at least 4 days.
15 g is dispensed if you fully depress the actuator for approximately 1 minute. Spraying for 2 seconds provides approximately 0.5 g of Enstilar. As a guide, 0.5 g of foam should cover an area of skin roughly corresponding to the surface area of an adult hand
• avoid use on more than 30% of your body
• avoid use on large areas of damaged skin, on mucous membranes or in skin folds (groin, armpits, under breasts) as this increases the absorption of the steroid
• avoid use on the face or on genitals (sexual organs) as they are very sensitive to steroids
• avoid excessive sunbathing, excessive use of a solarium and other forms of light treatment, as your skin is sensitive to light.
Children and adolescents
Enstilar is not recommended for the use in children below the age of 18 years.
Other medicines and Enstilar
Tell your doctor, pharmacist or nurse if you are using, have recently used, or might use any other medicines.
Pregnancy and breast-feeding
If you are pregnant or breast-feeding, think you may be pregnant or are planning to have a baby, ask your doctor for advice before using this medicine.
If your doctor has agreed that you can breast-feed, take care and do not apply Enstilarto the breast area.
See ‘Instruction for proper use’.
Driving and using machines
This medicine should not have any effect on your ability to drive or use machines.
Enstilar contains butylhydroxytoluene (E321)
This may cause local skin reactions (e.g. contact dermatitis), or irritation to the eyes and mucous membranes.
See ‘Instruction for proper use’.
3. How to use Enstilar®
Always use this medicine exactly as your doctor has told you. Check with your doctor or pharmacist if you are not sure.
Enstilar is for cutaneous use (on the skin).
Before use, read the patient information, even if you have used Enstilar before.
Enstilar is designed for direct application (spray-on) to your skin affected by psoriasis vulgaris.
Instruction for proper use
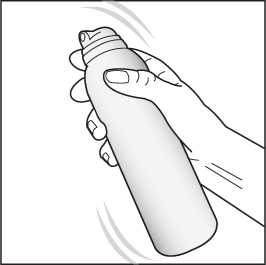
1. Shake the can for a few seconds before use.
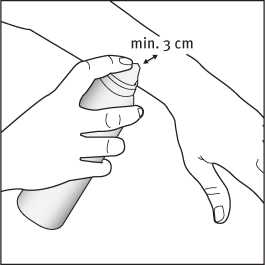
2. Apply the foam by holding the can at least 3 cm from the skin and spray directly onto each affected area.
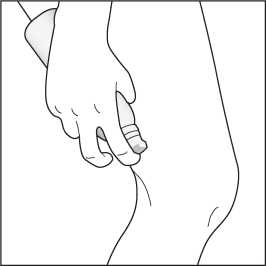
3. The foam can be sprayed holding the can in any orientation except horizontally.
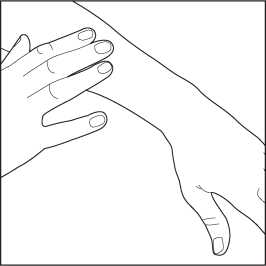
4. Gently rub the foam into each affected skin area.
5. After applying the foam, put the cap back on the can to prevent accidental spraying when not in use.
6. Wash your hands well after using Enstilar (unless you are using the foam to treat your hands). This will avoid accidentally spreading the foam to other parts of your body (especially the face, mouth and eyes).
Further information for proper use:
• use only on your psoriasis and do not use on skin which does not have psoriasis
• wash or rinse well if, by accident, you have applied foam to your eyes, mouth, sexual organs or breasts if you are breast-feeding
• do not worry if some foam accidentally gets on normal skin near your psoriasis, but wipe it off if it spreads too far
• do not bandage, tightly cover or wrap the treated skin area
• in order to achieve optimal effect, it is recommended not to take a shower or bath immediately after spraying the foam
• after applying the foam, avoid contact with textiles which are easily stained by grease (e.g. silk).
Duration of treatment
• apply the foam once daily. It may be more convenient to use the foam in the evening.
• the normal treatment period is 4 weeks but your doctor may decide on a different treatment period.
If you have used more Enstilar than you should Important: One 60 g can of Enstilar should last for at least 4 days (see section 2 ’Special precautions’). If you use other calcipotriol containing medicines, the total amount of calcipotriol medicines, including Enstilar, must not exceed 15 grams per day. Contact your doctor if you have used more than the recommended dose.
Excessive use of Enstilar may cause a problem with calcium in your blood, which usually returns to normal when treatment is discontinued.
Excessive prolonged use can also cause your adrenal glands to stop working properly (the adrenal glands are found near the kidneys and produce hormones).
See section 4 for further information.
If you forget to use Enstilar
Do not use a double dose to make up for forgotten doses.
If you stop using Enstilar
The use of Enstilar should be stopped as indicated by your doctor. It may be necessary for you to stop this medicine gradually, especially if you have used it for a long time.
If you have any further questions about the use of this medicine, ask your doctor, pharmacist or nurse.
4. Possible side effects
Like all medicines, this medicine can cause side effects, although not everybody gets them.
Side effects that have been observed for Enstilar:
Uncommon side effects (may affect up to 1 in 100 people):
• allergic reaction. Symptoms may include skin rash and swelling of the skin, face or mouth
• worsening or ‘flare up’ of your psoriasis after treatment has stopped
• a rise in calcium levels in the blood
• skin irritation which might include itching, burning and stinging and/or redness of the skin
• redness, pain or swelling of the hair roots of the skin (folliculitis)
• loss of skin colour (depigmentation).
Enstilar contains betamethasone (a strong steroid) and calcipotriol. You may therefore experience the following side effects. These side effects are more likely to happen if Enstilar is used for a long time, if used under dressings or in skin folds (e.g. groin, armpits or under breasts), or if used on large skin areas:
• allergic reactions with swelling of the face or other parts of the body such as the hands or feet. Swelling of the mouth/throat and trouble breathing may also occur
• calcium levels in your blood or urine may increase so much that you get symptoms. Signs are frequent urination, constipation, muscle weakness, and confusion. When the treatment is stopped, the calcium levels return to normal
• your adrenal glands may stop working properly. Signs are tiredness, depression, anxiety
• cloudy vision, difficulty seeing at night, sensitivity to light (this could be a sign of cataracts)
• eye pain, red eye, decreased or cloudy vision (this could be a sign of increased pressure inside the eye)
• infections (because your immune system is weakened)
• pustular psoriasis (a red area of psoriasis with yellowish pustules (pimples))
• you may experience fluctuations in blood sugar levels.
If you experience any of the above side effects you should contact your doctor immediately.
Less serious side effects known to be caused by calcipotriol or betamethasone include the following:
• thinning of the skin
• stretch marks
• blood vessels under your skin may become more noticable
• changes in hair growth
• red rash around the mouth (perioral dermatitis)
• worsening of your psoriasis
• sensitivity of the skin to light resulting in a rash
• itchy skin rash (eczema)
• white or grey hair can transiently change to a yellowish colour at the application site when used on the scalp.
Reporting of side effects
If you get any side effects, talk to your doctor, pharmacist or nurse. This includes any possible side effects not listed in this leaflet. You can also report side effects directly via the national reporting system.
For Ireland, via HPRA Pharmacovigilance,
Earlsfort Terrace, IRL — Dublin 2 Tel: +353 1 6764971 Fax: +353 1 6762517 Website: www.hpra.ie E-mail: medsafety@hpra.ie
For the United Kingdom, via the Yellow Card Scheme, Website: www.mhra.gov.uk/yellowcard By reporting side effects you can help provide more information on the safety of this medicine.
5. How to store Enstilar®
Caution: Extremely flammable aerosol. Pressurised container: May burst if heated. Protect from sunlight. Do not expose to temperatures exceeding 50°C. Do not pierce or burn, even after use. Do not spray on an open flame or other ignition source. Keep away from sparks, open flames and other ignition sources. No smoking near the can.
Keep this medicine out of the sight and reach of children.
Do not use Enstilar after the expiry date, which is stated on the carton and can after EXP. The expiry date refers to the last day of that month.
Do not store above 30°C.
The can should be discarded 6 months after first opening. Medicines should not be disposed of via wastewater or household waste. Ask your pharmacist how to dispose of medicines no longer required. These measures will help to protect the environment.
6.Contents of the pack and other information What Enstilar contains
The active substances are:
Calcipotriol and betamethasone.
One gram of cutaneous foam contains 50 micrograms of calcipotriol (as monohydrate) and 0.5 mg of betamethasone (as dipropionate).
The excipients are:
Liquid paraffin
Polyoxypropylene stearyl ether All-rac-a-tocopherol White soft paraffin Butylhydroxytoluene (E321)
Butane
Dimethyl ether.
What Enstilar looks like and contents of the pack
Enstilar is a cutaneous foam.
After spraying, a white to off-white foam is formed. Aluminium can with a polyamide-imide inner lacquer, equipped with a continuous valve and actuator.
The can contains 60 g of foam, not including the amount of propellants.
Pack sizes: 60 g, 2 x 60 g.
Not all pack sizes may be marketed.
Marketing Authorisation Holder and Manufacturer Marketing Authorisation Holder
LEO Pharma A/S Industriparken 55 DK-2750 Ballerup Denmark
Manufacturer
Colep Laupheim GmbH & Co. KG FockestraGe 12 DE-88471 Laupheim Germany
This medicinal product is authorised in the Member States of the EEA under the following names:
Enstilar: Austria, Bulgaria, Croatia, Cyprus, Czech Republic, Denmark, Estonia, Finland, France, Germany, Greece, Hungary, Iceland, Ireland, Italy, Latvia, Lithuania, Malta, Netherlands, Norway, Poland, Portugal, Slovak Republic, Spain, Sweden, United Kingdom Enstilum: Belgium, Luxembourg, Romania, Slovenia
This leaflet was last revised in March 2016.
Detailed information on this medicine is available on the website of the Health Products Regulatory Authority, www.hpra.ie. and Medicines and Healthcare products Regulatory Agency, www.mhra.gov.uk.
LEO 050522
ENSTILAR- calcipotriene monohydrate and betamethasone dipropionate aerosol, foam
LEO Pharma Inc.
1 INDICATIONS AND USAGE
Enstilar® (calcipotriene and betamethasone dipropionate) Foam is indicated for the topical treatment of plaque psoriasis in patients 12 years and older.
2 DOSAGE AND ADMINISTRATION
Shake can prior to using Enstilar Foam. Apply Enstilar Foam to affected areas once daily for up to 4 weeks. The maximum dose should not exceed 60 grams every 4 days. Rub in Enstilar Foam gently. Wash hands after applying the product. Discontinue Enstilar Foam when control is achieved.
Enstilar Foam should not be used:
- with occlusive dressings unless directed by a healthcare provider.
- on the face, groin, or axillae, or if skin atrophy is present at the treatment site.
Enstilar Foam is not for oral, ophthalmic, or intravaginal use.
3 DOSAGE FORMS AND STRENGTHS
Enstilar Foam: 0.005%/0.064% — each gram contains 50 mcg calcipotriene and 0.643 mg of betamethasone dipropionate in a white to off-white opalescent liquid in a pressurized aluminum spray can with a continuous valve and actuator. At administration the product is a white to off-white foam after evaporation of the propellants.
4 CONTRAINDICATIONS
None.
5 WARNINGS AND PRECAUTIONS
5.1 Flammability
The propellants in Enstilar Foam are flammable. Instruct the patient to avoid fire, flame, and smoking during and immediately following application.
5.2 Hypercalcemia and Hypercalciuria
Hypercalcemia and hypercalciuria have been observed with use of Enstilar Foam. If hypercalcemia or hypercalciuria develop, discontinue treatment until parameters of calcium metabolism have normalized. The incidence of hypercalcemia and hypercalciuria following Enstilar Foam treatment of more than 56 weeks has not been evaluated [ see Clinical Pharmacology (12.2)].
5.3 Effects on Endocrine System
Hypothalamic-Pituitary-Adrenal Axis Suppression
Systemic absorption of topical corticosteroids can cause reversible hypothalamic-pituitary-adrenal (HPA) axis suppression with the potential for clinical glucocorticosteroid insufficiency. This may occur during treatment or upon withdrawal of treatment. Factors that predispose a patient to HPA axis suppression include the use of high-potency steroids, large treatment surface areas, prolonged use, use of occlusive dressings, altered skin barrier, liver failure, and young age.
Evaluation for HPA axis suppression may be done by using the adrenocorticotropic hormone (ACTH) stimulation test. If HPA axis suppression is documented, gradually withdraw Enstilar Foam, reduce the frequency of application, or substitute with a less potent corticosteroid.
The following trials evaluated the effects of Enstilar Foam on HPA axis suppression [see Clinical Pharmacology (12.2) ]:
- In a trial evaluating the effects of Enstilar Foam on the HPA axis, 35 adult subjects applied Enstilar Foam on the body and scalp. Adrenal suppression was not observed in any subjects after 4 weeks of treatment.
- In another trial, 33 adolescent subjects age 12 to 17 years applied Enstilar Foam on the body and scalp. Adrenal suppression occurred in 3 (9%) of the subjects.
- In a trial, 21 subjects aged 18 years and older with plaque psoriasis applied Enstilar Foam once daily for 4 weeks and then twice weekly on 2 non-consecutive days for 52 weeks, including once daily for 4 weeks if loss of response occurred. Adrenal suppression was observed in 2 (10%) of the subjects.
Cushing’s Syndrome and Hyperglycemia
Systemic effects of topical corticosteroids may also include Cushing’s syndrome, hyperglycemia, and glucosuria.
Additional Considerations for Endocrine Adverse Reactions
Pediatric patients may be more susceptible to systemic toxicity due to their larger skin surface to body mass ratios [see Use in Specific Populations (8.4)].
Use of more than one corticosteroid-containing product at the same time may increase the total systemic corticosteroid exposure.
5.4 Allergic Contact Dermatitis
Allergic contact dermatitis has been observed with topical calcipotriene and topical corticosteroids. Allergic contact dermatitis to a topical corticosteroid is usually diagnosed by observing a failure to heal rather than a clinical exacerbation. Corroborate such an observation with appropriate diagnostic patch testing.
5.5 Ophthalmic Adverse Reactions
Use of topical corticosteroids, including Enstilar® Foam, may increase the risk of posterior subcapsular cataracts and glaucoma. Cataracts and glaucoma have been reported with the postmarketing use of topical corticosteroid products. Avoid contact with Enstilar Foam with eyes. Enstilar Foam may cause eye irritation. Advise patients to report any visual symptoms and consider referral to an ophthalmologist for evaluation.
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Clinical Trials Conducted in Subjects 18 years and older with Psoriasis
The rates of adverse reactions described below were from three randomized, multicenter, vehicle and/or active-controlled clinical trials in adult subjects with plaque psoriasis [see Clinical Studies (14)]. Subjects applied study product once daily for 4 weeks, and the median weekly dose of Enstilar Foam was 25 grams. Adverse reactions reported in <1% of adult subjects treated with Enstilar Foam included: application site irritation, application site pruritus, folliculitis, skin hypopigmentation, hypercalcemia, urticaria, and exacerbation of psoriasis.
Clinical Trials Conducted in Subjects 12 to 17 years with Psoriasis
In one uncontrolled clinical trial, 106 subjects aged 12 to 17 years with plaque psoriasis of the scalp and body applied Enstilar Foam once daily for up to 4 weeks. The median weekly dose was 40 grams. Adverse reactions reported in <1% of adolescent subjects treated were acne, erythema, application site pain, and skin reactions [see Use in Specific Populations (8.4) and Clinical Pharmacology (12.2)].
6.2 Postmarketing Experience
Because adverse reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Postmarketing reports for local adverse reactions to Enstilar Foam included application site burning.
Postmarketing reports for local adverse reactions to topical corticosteroids included atrophy, striae, telangiectasia, dryness, perioral dermatitis, secondary infection, and miliaria.
Ophthalmic adverse reactions of cataracts, glaucoma, and increased intraocular pressure have been reported with the use of topical corticosteroids, including topical betamethasone products.
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
Available data with Enstilar Foam are not sufficient to evaluate a drug-associated risk for major birth defects, miscarriages, or adverse maternal or fetal outcomes. Although there are no available data on use of the calcipotriene component in pregnant women, systemic exposure to calcipotriene after topical administration of Enstilar Foam is likely to be low [see Clinical Pharmacology (12.3)].
Observational studies suggest an increased risk of having low birth weight infants with the maternal use of potent or super potent topical corticosteroids (see Data). Advise pregnant women that Enstilar® Foam may increase the potential risk of having a low birth weight infant and to use Enstilar Foam on the smallest area of skin and for the shortest duration possible.
In animal reproduction studies, oral administration of calcipotriene to pregnant rats during the period of organogenesis resulted in an increased incidence of minor skeletal abnormalities, including enlarged fontanelles and extra ribs (see Data). Oral administration of calcipotriene to pregnant rabbits during the period of organogenesis had no apparent effects on embryo-fetal development. Subcutaneous administration of betamethasone dipropionate to pregnant rats and rabbits during the period of organogenesis resulted in fetal toxicity, including fetal deaths, reduced fetal weight, and fetal malformations (cleft palate and crooked or short tail) (see Data). The available data do not allow the calculation of relevant comparisons between the systemic exposures of calcipotriene and betamethasone dipropionate observed in animal studies to the systemic exposures that would be expected in humans after topical use of Enstilar Foam.
The background risk of major birth defects and miscarriage in the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Data
Human Data
Available observational studies in pregnant women did not identify a drug-associated risk of major birth defects, preterm delivery, or fetal mortality with the use of topical corticosteroids of any potency. However, when the dispensed amount of potent or super potent topical corticosteroids exceeded 300 grams during the entire pregnancy, maternal use was associated with an increased risk of low birth weight in infants.
Animal Data
Embryo-fetal development studies with calcipotriene were performed by the oral route in rats and rabbits. Pregnant rats received dosages of 0, 6, 18, or 54 mcg/kg/day (0, 36, 108, and 324 mcg/m2 /day, respectively) on days 6-15 of gestation (the period of organogenesis). There were no apparent effects on maternal survival, behavior, or body weight gain, no effects on litter parameters, and no effects on the incidence of major malformations in fetuses. Fetuses from dams dosed at 54 mcg/kg/day exhibited a significantly increased incidence of minor skeletal abnormalities, including enlarged fontanelles and extra ribs.
Pregnant rabbits were dosed daily with calcipotriene at exposures of 0, 4, 12, or 36 mcg/kg/day (0, 48, 144, and 432 mcg/m2 /day, respectively) on days 6-18 of gestation (the period of organogenesis). Mean maternal body weight gain was reduced in animals dosed at 12 or 36 mcg/kg/day. The incidence of fetal deaths was increased in the group dosed at 36 mcg/kg/day; reduced fetal weight was also observed in this group. The incidence of major malformations among fetuses was not affected. An increase in the incidence of minor skeletal abnormalities, including incomplete ossification of sternebrae, pubic bones, and forelimb phalanges, was observed in the group dosed at 36 mcg/kg/day.
Embryo-fetal development studies with betamethasone dipropionate were performed via subcutaneous injection in mice and rabbits. Pregnant mice were administered doses of 0, 156, 625, or 2500 mcg/kg/day (0, 468, 1875, and 7500 mcg/m2 /day, respectively) on days 7 through 13 of gestation (the period of organogenesis). Betamethasone dipropionate induced fetal toxicity, including fetal deaths, reduced fetal weight, malformations (increased incidence of the cleft palate and crooked or short tail), and minor skeletal abnormalities (delayed ossification of vertebra and sternebrae). Fetal toxicity was observed at the lowest exposure that was evaluated (156 mcg/kg/day).
Pregnant rabbits were injected subcutaneously at dosages of 0, 0.625, 2.5, and 10 mcg/kg/day (0, 7.5, 30, and 120 mcg/m2 /day, respectively) on days 6 through 18 of gestation (the period of organogenesis). Betamethasone dipropionate induced fetal toxicity, including fetal deaths, reduced fetal weight, external malformations (including malformed ears, cleft palate, umbilical hernia, kinked tail, club foot, and club hand), and skeletal malformations (including absence of phalanges of the first digit and cranial dysplasia) at dosages of 2.5 mcg/kg/day and above.
Calcipotriene was evaluated for effects on peri- and post-natal development when orally administered to pregnant rats at dosages of 0, 6, 18 or 54 mcg/kg/day (0, 36, 108, and 324 mcg/m2 /day, respectively) from gestation day 15 through day 20 postpartum. No remarkable effects were observed on any parameter, including survival, behavior, body weight, litter parameters, or the ability to nurse or rear pups.
Betamethasone dipropionate was evaluated for effects on peri- and post-natal development when orally administered to pregnant rats at dosages of 0, 100, 300, and 1000 mcg/kg/day (0, 600, 1800, and 6000 mcg/m2 /day, respectively) from gestation day 6 through day 20 postpartum. Mean maternal body weight was significantly reduced on gestation day 20 in animals dosed at 300 and 1000 mcg/kg/day. The mean duration of gestation was slightly, but statistically significantly, increased at 100, 300, and 1000 mcg/kg/day. The mean percentage of pups that survived to day 4 was reduced in relation to dosage. On lactation day 5, the percentage of pups with a reflex to right themselves when placed on their back was significantly reduced at 1000 mcg/kg/day. No effects on the ability of pups to learn were observed, and the ability of the offspring of treated rats to reproduce was not affected.
RxDrugLabels.com provides trustworthy package insert and label information about marketed prescription drugs as submitted by manufacturers to the U.S. Food and Drug Administration. Package information is not reviewed or updated separately by RxDrugLabels.com. Every individual prescription drug label and package insert entry contains a unique identifier which can be used to secure further details directly from the U.S. National Institutes of Health and/or the FDA.
https://rxdruglabels.com/lib/rx/rx-meds/enstilar/
30.06.2016
В Великобритании выпущен препарат Энстилар в виде пены для лечения псориаза
На текущей неделе датская фармацевтическая компания Лео Фарма (LEO Pharma) сообщила, что в Великобритании выпущен ее препарат Энстилар/ кальципотриен 50 мкг/г / бетаметазона дипропионат 0,5 мг/г (Enstilar — calcipotriene/betamethasone dipropionate 50 micrograms/g / 0,5 mg/g) для лечения псориаза.
Данное средство в виде пены предствляет собой фиксированную комбинацию действующих веществ для применения один раз в день для местного лечения всех проявлений бляшечного псориаза у пациентов в возрасте от 18 лет. Согласно представленным данным Энстилар является более эффективным комбинированным средством для местного применения, чем другие доступные препараты. В целом Энстилар (кальципотриен + бетаметазона дипропионат) хорошо переносится, а у более половины пациентов из клинических испытаний было отмечено существенное улучшение видимых признаков заболевания через четыре недели применения, а некоторых — даже на первой неделе.
Выпуск препарата Энстилар (кальципотриен + бетаметазона дипропионат) в Великобритании стал возможен благодаря положительному рассмотрению результатов базового исследования PSO-FAST IIIa Фазы, в котором оценивались его эффективность и профиль безопасности в течение четырех недель, а также сведений испытания MUSE II Фазы по изучению профиля безопасности препарата. Читайте также: Компания Лео Фарма планирует зарегистрировать новую форму выпуска препарата Таклонекс для лечения бляшечного псориаза В клиническом исследовании PSO-FAST у более чем половины пациентов, которые получали лечение препаратом Энстилар, было достигнуто полное или практически полное очищение кожи на 4-ой неделе по шкале общей оценки ответа на лечение.
Кроме того, у более половины пациентов, которые получали лечение препаратом Энстилар (кальципотриен + бетаметазона дипропионат), было достигнуто 75%-ное улучшение по модифицированному индексу тяжести поражения псориазом (modified Psoriasis Area and Severity Index score, mPASI) от базовых показателей через четыре недели терапии.
Как прокомментировала Жеральдина Мерфи (Geraldine Murphy), управляющий директор компании Лео Фарма Великобритания и Ирландия (LEO Pharma UK & Ireland), выпуск средства Энстилар является важным достижением для ее компании, которая предана улучшению результатов лечения и качества жизни людей, которые живут с такими кожными заболеваниями, как, например, псориаз. Она отметила, что это первое в своем роде комбинированное средство, а его выпуск в виде пены предоставляет новый более удобный вариант лечения.
Материал подготовлен порталом МЕДФАРМКОННЕКТ: http://medpharmconnect.com/News/Pharma_RD_News/21758.htm
Вернуться к списку новостей
